
(See Figure 3.4 “The Structure of the Atom”.) Figure 3.4 “The Structure of the Atom.” Atoms have protons and neutrons in the centre, making the nucleus, while the electrons orbit the nucleus. The electrons are outside the nucleus and spend their time orbiting in space about the nucleus. The relatively massive protons and neutrons are collected in the centre of an atom, in a region called the nucleus of the atom (plural nuclei). Experiments by Ernest Rutherford in England in the 1910s pointed to a nuclear model of the atom. How are these particles arranged in atoms? They are not arranged at random. Table 3.7 Properties of the Three Subatomic Particles Name Table 3.7 “Properties of the Three Subatomic Particles” summarizes the properties of these three subatomic particles. We now know that all atoms of all elements are composed of electrons, protons, and (with one exception) neutrons. The neutron is a subatomic particle with about the same mass as a proton but no charge. The proton is a more massive (but still tiny) subatomic particle with a positive charge, represented as p +. Later, two larger particles were discovered. It is often represented as e −, with the right superscript showing the negative charge. The first part to be discovered was the electron, a tiny subatomic particle with a negative charge. These concepts form the basis of chemistry.Īlthough the word atom comes from a Greek word that means “indivisible,” we understand now that atoms themselves are composed of smaller parts called subatomic particles. Atoms combine in whole-number ratios to form compounds.Atoms of the same element are the same atoms of different elements are different.The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory, first stated by John Dalton, an English scientist, in 1808. 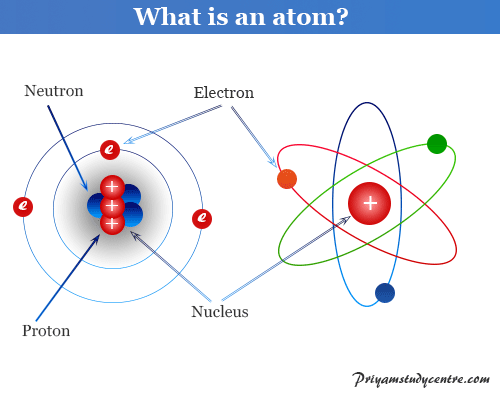
Atoms are so small that it is difficult to believe that all matter is made from atoms-but it is. The period at the end of a printed sentence has several million atoms in it. It would take about fifty million atoms in a row to make a line that is 1 cm long. More modern atomic theories have made some modifications, for example to include the existence of atomic isotopes, that is, atoms with different numbers of neutrons, but the same number of protons and electrons, and the conversion of energy into matter and vice versa, but Dalton’s core ideas remain valid.The smallest piece of an element that maintains the identity of that element is called an atom. Chemical reactions are due to the rearrangements of atoms, and atoms (matter) are neither created nor destroyed during a reaction.īased on these tenets he was able to explain many of the observations that had been made, by himself and others, about how matter behaves and reacts.Compounds are formed by combinations of atoms of two or more elements.Atoms of a given element are different from atoms of other elements.All atoms of an element are identical and have the same mass and properties.Elements are composed of small indivisible, indestructible particles called atoms.Dalton’s atomic theory (1805) had a number of important components: Among these rules was the observation that the total matter present in a system does not change during a chemical reaction, although a reaction might lead to a change from a solid to a gas or vice versa. Rather surprisingly, Dalton never really explained what led him to propose his atomic theory, although he certainly used it to explain existing rules about how different elements combine. In 1805 Dalton published his atomic theory to explain the observed law of multiple, or definite, proportions, which stated briefly is “when elements combine, they do so in the ratio of small whole numbers”, we will return to this idea later on, in much greater detail. The first scientific theory of atomic structure was proposed by John Dalton (1766–1844), a self-taught Quaker living in Manchester, England. That said, it was not until Albert Einstein (1874-1955) proposed his general theory of relativity in 1915 that there was a coherent, mechanistic explanation for gravitational forces. It requires a capacity for deep, original, and complex thought. The point is that Newton was able to make sense of the data, something that is in no way trivial. 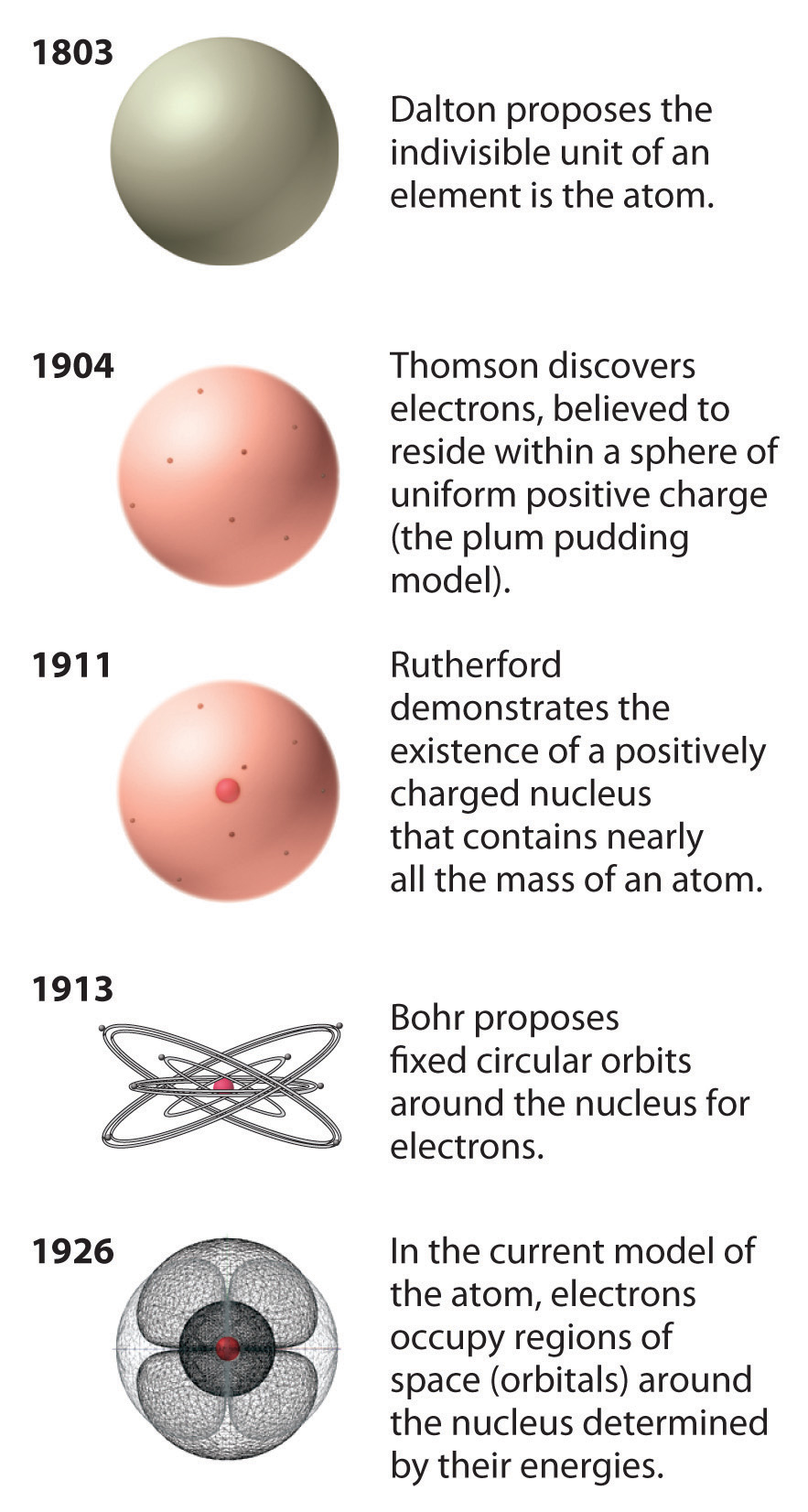
This seems like a pretty weird and rather over-blown speculation how does this “action at a distance” between two objects work? Yet, followed scientifically, it appeared to be very powerful and remarkably accurate. \) where \(r\) is the distance between two objects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
